 Most atoms do not have eight electrons in their valence electron shell. Use Lewis diagrams to illustrate ion formation.Covalent bonding and covalent compounds will be discussed in Chapter 4 "Covalent Bonding and Simple Molecular Compounds". The bond made by electron sharing is called a covalent bond. These shared electrons simultaneously occupy the outermost shell of more than one atom. The second way for an atom to obtain an octet of electrons is by sharing electrons with another atom. and are the primary subject of this chapter. The resulting compounds are called ionic compounds A compound formed with an ionic bond. Because opposite charges attract (while like charges repel), these oppositely charged ions attract each other, forming ionic bonds An attraction between oppositely charged ions. Charged atoms are called ions A charged atom. Those that lose electrons become positively charged, and those that gain electrons become negatively charged. Because some atoms will lose electrons and some atoms will gain electrons, there is no overall change in the number of electrons, but individual atoms acquire a nonzero electric charge. One way is the transfer of electrons between two atoms until all atoms have octets. There are two ways for an atom that does not have an octet of valence electrons to obtain an octet in its outer shell. Of the noble gases, only krypton, xenon, and radon have been found to make compounds. This useful rule of thumb is called the octet rule The idea that atoms tend to have eight electrons in their valence shell., and it is a key to understanding why compounds form. Chemists have concluded that atoms are especially stable if they have eight electrons in their outermost shell. 
What else do the noble gas elements have in common? Except for helium, they all have eight valence electrons. These elements-helium, neon, argon, krypton, xenon, and radon-do not form compounds very easily, which suggests that they are especially stable as lone atoms. What causes atoms to make a chemical bond with other atoms, rather than remaining as individual atoms? A clue comes by considering the noble gas elements, the rightmost column of the periodic table. Chemical bonds are formed when electrons in different atoms interact with each other to make an arrangement that is more stable than when the atoms are apart. We will see additional examples of such differences in this chapter and Chapter 4 "Covalent Bonding and Simple Molecular Compounds", as we consider how atoms combine to form compounds.Ītoms can join together by forming a chemical bond A very strong attraction between two atoms., which is a very strong attraction between two atoms. Nevertheless, the compound has properties completely different from either elemental sodium (a chemically reactive metal) or elemental chlorine (a poisonous, green gas). Table salt, as we have seen, consists of only two elements: sodium and chlorine. Compounds can be very complex combinations of atoms, but many important compounds are fairly simple. There are only 118 known chemical elements but tens of millions of known chemical compounds. Most salt substitutes use potassium instead of sodium, but some people complain that the potassium imparts a slightly bitter taste. 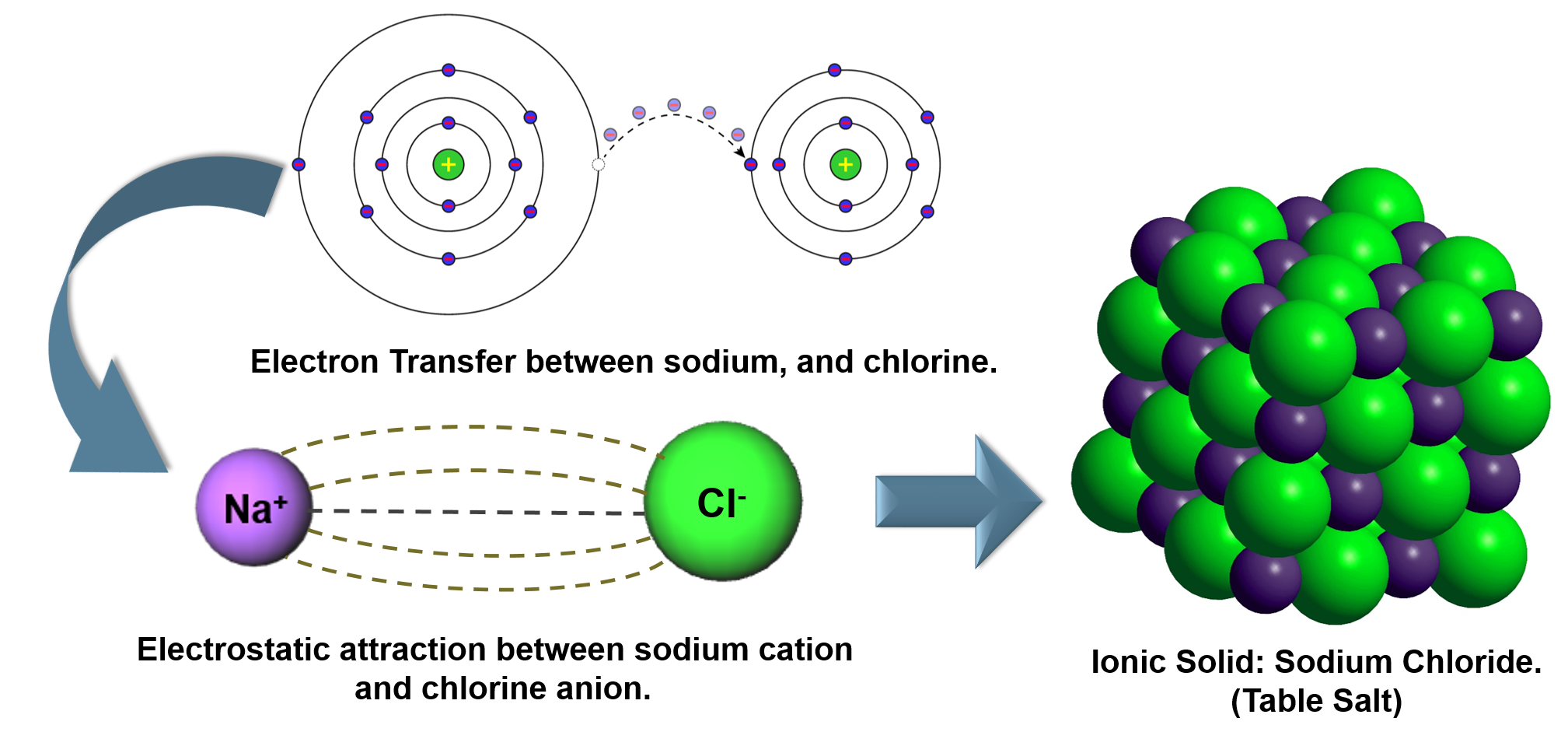
Even so, some doctors still recommend a low-salt diet (never a “no-salt” diet) for patients with high blood pressure, which may include using a salt substitute. Previously, the high ingestion of salt was thought to be associated with high blood pressure, but current research does not support this link. The RDI of sodium is 2,400 mg-the amount in about 1 teaspoon of salt-but the average intake of sodium in the United States is between 4,000 mg and 5,000 mg, partly because salt is a common additive in many prepared foods. In fact, human blood is about a 0.9% sodium chloride solution, and a solution called normal saline is commonly administered intravenously in hospitals.Īlthough some salt in our diets is necessary to replenish the sodium and chloride ions that we excrete in urine and sweat, too much is unhealthy, and many people may be ingesting more salt than their bodies need. 
Sodium, for example, is important for nerve conduction and fluid balance. Table salt is sodium chloride (NaCl), which is a simple compound of two elements that are necessary for the human body to function properly. Today, table salt is either mined or obtained from the evaporation of saltwater. 
Two thousand years ago, Roman soldiers received part of their pay as salt, which explains why the words salt and salary come from the same Latin root ( salarium). This kind of salt is used as a condiment throughout the world, but it was not always so abundant. We will see that the word salt has a specific meaning in chemistry, but to most people, this word refers to table salt. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
